 Occasionally, two molecules colliding head on at exactly the same speed would stop completely before being hit by another molecule and regaining some speed.) In liquids, the distance between collisions is very small, but the speeds are about the same as in a gas at the same temperature. They go only about 200 times their diameter before colliding violently with another molecule and losing or gaining energy. N 2 and O 2 molecules are at 298 K are gases, of course, and have a very wide range of speeds, from zero to more than two thousand miles an hour with an average of roughly a thousand miles an hour. 
In other words, the system - the macrostate - rapidly and successively changes to be in a gigantic number of different microstates out of the “gazillions” of accessible microstates, (In solids, the location of the particles is almost the same from instant to instant, but not exactly, because the particles are vibrating a tiny amount from a fixed point at enormous speeds.) Every microstate for a particular system has exactly the total energy of the macrostate because a microstate is just an instantaneous quantum energy-photo of the whole system.) That's why, in an instant for any particular macrostate, its motional energy* has been rearranged as to what molecule has what amount of energy. (The total energy hasn't changed when molecular movement changes one microstate into another. But if only one molecule moves a bit slower because it had hit another and made that other one move an exactly equal amount faster - then that would be a different microstate. In the next instant - and that really means in an extremely short time - at least a couple of moving molecules out of the 6 x 10 23 will hit one another. That's why it is wise to talk in terms of “an instant”!) To take a photo like that may seem impossible and it is. Considering a mole of molecules (6 x 10 23) traveling at a very large number of different speeds, the collisions occur - and thus changes in energy of trillions of molecules occurs - in far less than a trillionth of a second. (A molecule moving at an average speed of around a thousand miles an hour collides with others about seven times in a billionth of a second. That is a more modern view that we will use.) In the next instant the system immediately changes to another microstate. In quantum mechanics the behavior of molecules is only described in terms of their energies on particular energy levels. (This is talking in ‘classical mechanics’ language where molecules are assumed to have location and momentum. One microstate then is something like a theoretical "absolutely instantaneous photo" of the location and momentum of each molecule and atom in the whole macrostate. Each specific way, each arrangement of the energy of each molecule in the whole system at one instant is called a microstate." In liquids and gases, the particles themselves are constantly redistributing in location as well as changing in the quanta (the individual amount of energy that each molecule has) due to their incessantly colliding, bouncing off each other with (usually) a different amount of energy for each molecule after the collision. "In a system its energy is constantly being redistributed among its particles. In contrast, a microstate for a system is all about time and the energy of the molecules in that system. 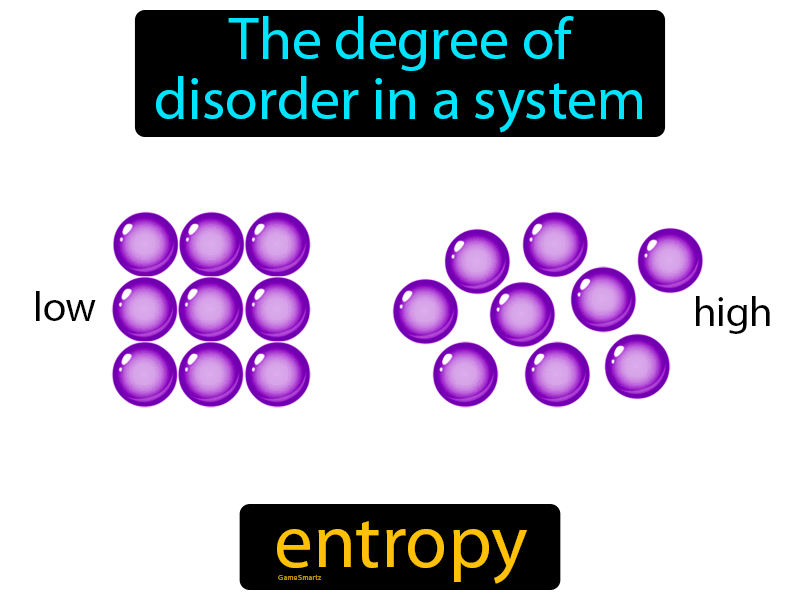 
Thus, a macrostate does not change over time if its observable properties do not change. In calculations involving entropy, the ΔH of any phase change in a substance (“phase change energy”) is added to motional energy, but it is unaltered in ordinary entropy change (of heating, expansion, reaction, etc.) unless the phase itself is changed.Ī macrostate is the thermodynamic state of any system that is exactly characterized by measurement of the system's properties such as P, V, T, H and number of moles of each constituent. *Motional energy includes the translational, rotational, and vibrational modes of molecular motion. But in thermodynamics, a microstate isn't just about a smaller amount of matter', it is a detailed look at the energy that molecules or other particles have.) A microstate is one of the huge number of different accessible arrangements of the molecules' motional energy* for a particular macrostate. (Admittedly, a macrostate always has to involve an amount of matter large enough for us to measure its volume or pressure or temperature, i.e. Instead, they are two very different ways of looking at a system. \)ĭictionaries define “macro” as large and “micro” as very small but a macrostate and a microstate in thermodynamics aren't just definitions of big and little sizes of chemical systems. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
